Water, in its various forms, is an indispensable resource for life, industry, and technology. From cooling vast data centers to generating electricity and regulating environmental conditions, its unique thermophysical properties make it an unparalleled medium for energy transfer. Among these properties, the “heat of vaporization” stands out as a foundational concept, dictating how much energy is required to transform liquid water into steam. For engineers, designers, and innovators across the technology spectrum, understanding and harnessing this specific energy dynamic is not merely academic; it is an imperative for efficiency, sustainability, and breakthrough innovation.

This article delves into the technological significance of the heat of vaporization of water. We will explore its definition, quantify its value in practical terms, and critically examine its diverse applications in technology. We will discuss how this property influences the design of critical systems, the pursuit of energy efficiency, and the development of future technologies that rely on advanced thermal management.
The Core Concept: Bridging Physics to Practical Tech
At its heart, the heat of vaporization is a fundamental thermodynamic property. However, its translation into tangible technological solutions requires a deep understanding of its nuances and implications. For engineers, it represents a potent tool—or a significant challenge—in managing energy.
Defining Latent Heat and Phase Change
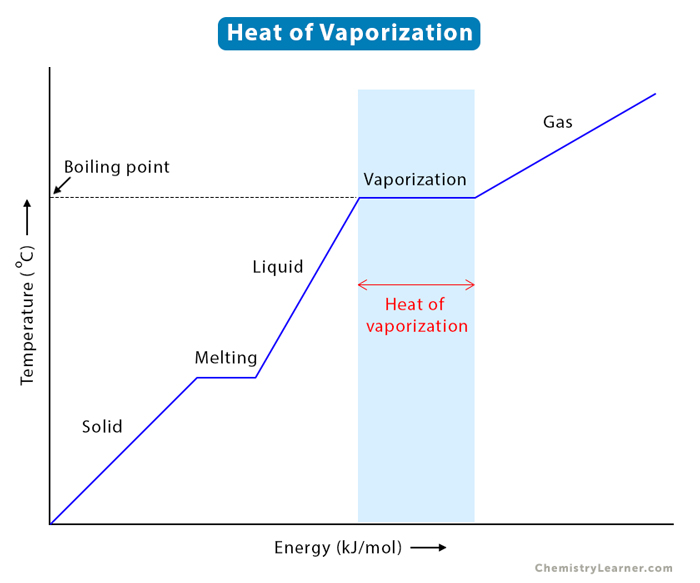
The heat of vaporization, often referred to as the enthalpy of vaporization or latent heat of vaporization, is the amount of energy absorbed by a substance when it changes from a liquid to a gas at constant temperature and pressure. For water, this process occurs during boiling or evaporation. Unlike sensible heat, which causes a change in temperature, latent heat facilitates a change in phase without an immediate temperature increase. This characteristic is precisely what makes water so valuable in many technological applications. When water evaporates, it absorbs a substantial amount of energy from its surroundings, effectively cooling them down. Conversely, when steam condenses back into liquid water, it releases that same amount of latent heat, which can then be captured and utilized for heating or power generation.
In technological systems, this phase change mechanism is a cornerstone. Think of a refrigeration cycle: a refrigerant (often a derivative of water or a similar fluid) absorbs heat from a space by evaporating, then releases that heat elsewhere by condensing. For water, this principle is scaled up and applied in myriad ways, from large-scale industrial cooling towers to sophisticated heat pipes in electronics. The efficiency of these systems hinges directly on how effectively the latent heat of vaporization can be managed and transferred.
The Specific Value for Water and its Uniqueness
Water possesses an exceptionally high heat of vaporization compared to many other common substances. At atmospheric pressure (101.325 kPa) and its boiling point (100°C or 212°F), the heat of vaporization of water is approximately 2257 kJ/kg (kilojoules per kilogram), or 540 cal/g (calories per gram). To put this into perspective, it takes roughly 4.18 kJ to raise the temperature of 1 kg of water by 1°C. This means that converting 1 kg of water to steam at 100°C requires approximately 540 times more energy than heating that same kilogram of water from 99°C to 100°C.
This high value is largely due to the strong hydrogen bonds between water molecules. Overcoming these intermolecular forces requires significant energy input. This unique property makes water an incredibly efficient medium for:
- Heat absorption: It can absorb a vast amount of thermal energy without experiencing a large temperature increase during evaporation, making it excellent for cooling.
- Energy storage and transfer: Steam carries a tremendous amount of energy, which can be transported and then released wherever heating or mechanical work is required.
- Thermal stability: Its ability to absorb latent heat provides a buffer against rapid temperature fluctuations, crucial for systems requiring stable thermal environments.
Beyond Boiling Point: Pressure and Purity Considerations
While the standard value for the heat of vaporization is typically cited at 100°C and atmospheric pressure, engineers constantly work with water under a wide range of conditions. The heat of vaporization is not a static value; it is dependent on both temperature and pressure. As temperature increases and pressure rises, the density difference between liquid and vapor phases diminishes, and consequently, the heat of vaporization decreases. This continues until the critical point is reached, where the distinction between liquid and gas phases disappears entirely, and the heat of vaporization becomes zero.
For instance, in high-pressure steam power plants, water is superheated far beyond its atmospheric boiling point, and the properties of steam (including its latent heat) are different. Engineers must use steam tables or advanced thermodynamic software to accurately determine these values for precise system design. Furthermore, the purity of water is crucial. Impurities can alter boiling points and heat transfer coefficients, affecting the efficiency and lifespan of technological systems. Therefore, in critical applications like power generation or semiconductor manufacturing, highly purified deionized water is often used to ensure predictable and optimal performance.
Technological Applications: Harnessing Water’s Energy Dynamics
The high heat of vaporization of water is not merely a scientific curiosity; it is a cornerstone for numerous technological advancements that underpin modern infrastructure and daily life. Its capacity to absorb and release large amounts of energy during phase change is exploited across diverse sectors.
HVAC and Refrigeration Systems: Cooling with Evaporation
One of the most intuitive applications of water’s heat of vaporization is in heating, ventilation, and air conditioning (HVAC) systems. Evaporative coolers, or “swamp coolers,” draw warm, dry air over water-saturated pads. As water evaporates from these pads, it absorbs latent heat from the air, causing the air temperature to drop significantly. While less common in humid climates, this technology is highly effective and energy-efficient in arid regions.
Beyond direct evaporative cooling, water-based chilled water systems are the backbone of large-scale air conditioning for commercial buildings and industrial facilities. Water is chilled by refrigerant cycles (which themselves often rely on latent heat principles) and then circulated through coils to absorb sensible heat from the air. Cooling towers, an essential component of many HVAC and industrial cooling systems, leverage the heat of vaporization on a grand scale. Warm water from the system is sprayed into a current of air; a small fraction of the water evaporates, carrying away a large amount of latent heat and cooling the remaining bulk water, which is then recirculated. This process is remarkably efficient in rejecting waste heat to the atmosphere.
Power Generation: Driving Turbines with Superheated Steam
The most prominent large-scale technological application of water’s heat of vaporization is in power generation. Thermal power plants—whether fossil fuel, nuclear, or concentrated solar—all operate on the same fundamental principle: heat is used to boil water, creating high-pressure, superheated steam. This steam then expands through turbines, converting thermal energy into mechanical energy, which drives generators to produce electricity.
The immense latent heat stored within steam means that a relatively small volume of water, once vaporized, can carry a tremendous amount of energy. This energy density makes steam an ideal working fluid for power cycles. The efficiency of these power plants, described by cycles like the Rankine cycle, is intricately linked to the precise control and utilization of water’s phase change properties at various temperatures and pressures. Engineers continually optimize boiler designs, turbine stages, and condenser systems to maximize the extraction of energy from this latent heat, pushing the boundaries of thermal efficiency.
Industrial Processes: Distillation, Drying, and Heat Transfer
In countless industrial processes, water’s heat of vaporization is a critical factor.
- Distillation: In chemical and petrochemical industries, distillation is used to separate components of liquid mixtures based on their boiling points. Water often serves as the heating medium (steam) to vaporize the mixture, and its condensation is used for cooling to re-condense the purified components. The energy required for vaporization of the target substance is a direct cost driver, making efficient heat recovery crucial.
- Drying: Industries from food processing to textiles and pharmaceuticals utilize drying processes that rely on evaporating water from products. The energy cost of removing water via vaporization is substantial, prompting the development of advanced drying technologies like vacuum drying or freeze-drying, which manipulate pressure to reduce the required latent heat or avoid direct vaporization.
- Heat Exchangers: Water, particularly as steam, is widely used as a heat transfer fluid in heat exchangers. Its high latent heat allows for efficient transfer of large amounts of energy within a compact footprint, making it invaluable for chemical reactors, pasteurization, and other processes requiring precise temperature control and efficient energy exchange.
Advanced Cooling Solutions: From Data Centers to Electronics
With the increasing miniaturization and power density of electronics, thermal management has become a critical engineering challenge. Water’s heat of vaporization offers potent solutions:
- Data Center Cooling: Hyperscale data centers consume vast amounts of energy, a significant portion of which is dissipated as heat. Immersion cooling, where servers are submerged in dielectric fluid that boils at low temperatures, is an emerging technology. However, even traditional liquid cooling systems often use water as the primary heat transfer medium, leveraging its high specific heat capacity and, in some cases, its latent heat through technologies like two-phase cooling or heat pipes/vapor chambers.
- Heat Pipes and Vapor Chambers: These passive heat transfer devices exploit the heat of vaporization and condensation. A working fluid (often water for suitable temperature ranges) evaporates at a hot end, absorbs latent heat, travels as vapor to a cooler end, condenses, releases latent heat, and returns as liquid via capillary action or gravity. They are integral to cooling laptops, smartphones, and high-performance computing components where efficient heat spreading is paramount.
- Direct Chip Liquid Cooling: As chip power densities continue to rise, direct-to-chip liquid cooling systems are becoming more prevalent. These systems circulate water (or other coolants) directly over or through microchannels embedded in the chip, effectively removing heat. Future advancements may involve phase-change cooling at the chip level, directly utilizing water’s latent heat for ultra-efficient thermal management.
Design and Optimization: Leveraging Vaporization for Efficiency
The practical application of the heat of vaporization of water in technology is fundamentally about engineering design and optimization. Maximizing efficiency, minimizing energy consumption, and ensuring reliability are paramount objectives driven by a deep understanding of this property.
Material Selection and System Design
Designing systems that effectively utilize water’s heat of vaporization involves careful material selection and intricate system architectures. Materials must withstand the temperatures and pressures associated with boiling and condensing water, resist corrosion, and possess good thermal conductivity. For example, in heat exchangers, the choice of metals (like copper or stainless steel) is crucial for efficient heat transfer. In power plants, specialized alloys are required for high-pressure, high-temperature steam applications.
System design also focuses on optimizing flow paths, surface areas for heat exchange, and the integration of components like pumps, valves, and condensers. For instance, in a cooling tower, the design of the fill material and spray nozzles directly impacts the rate of water evaporation and thus the cooling efficiency. In steam turbines, the geometry of the blades is meticulously engineered to efficiently extract mechanical energy from the expanding steam.
Energy Efficiency and Sustainability in Design
With growing environmental concerns and the drive for sustainability, leveraging the heat of vaporization of water for energy efficiency is a major focus. Engineers design systems to:
- Recover Waste Heat: Technologies like heat recovery steam generators (HRSG) capture waste heat from industrial processes or gas turbine exhausts to produce steam, which can then be used for power generation or process heating. This significantly improves overall energy utilization.
- Improve Coefficient of Performance (COP): In refrigeration and heat pump systems, the goal is to maximize the COP, which is the ratio of heating/cooling output to energy input. By optimizing the phase change cycle, engineers can achieve higher COPs, reducing electricity consumption.
- Minimize Water Consumption: While water is efficient for heat transfer, its evaporation in cooling towers leads to water loss. Engineers are developing “dry cooling” alternatives or hybrid systems that combine evaporative cooling with air cooling to conserve water, especially in water-stressed regions.
Predictive Modeling and Simulation Tools
Modern engineering relies heavily on advanced computational tools to model and simulate the behavior of systems involving water’s phase change. Computational Fluid Dynamics (CFD) software, thermodynamic modeling packages, and specialized heat transfer simulation tools allow engineers to:
- Predict Performance: Accurately forecast how a system will behave under various operating conditions, including transient effects.
- Optimize Designs: Experiment with different geometries, materials, and operating parameters virtually, reducing the need for expensive physical prototypes.
- Troubleshoot Issues: Identify potential problems like hot spots, cavitation, or inefficient heat transfer before they occur in the field.
These tools are indispensable for designing everything from micro-scale heat pipes to mega-scale power plants, ensuring that the immense energy potential of water’s heat of vaporization is harnessed with maximum precision and efficiency.
Emerging Technologies and Future Implications
The principles underlying water’s heat of vaporization will continue to drive innovation in emerging technologies, particularly those focused on sustainable energy, resource management, and advanced thermal solutions.
Water-Based Energy Storage
The concept of using water’s latent heat for energy storage is gaining traction. Just as a phase change material (PCM) can store and release large amounts of energy during its melting and freezing, water can store significant thermal energy during its vaporization and condensation. Systems are being explored where excess renewable energy (e.g., from solar or wind) is used to generate steam, which is then stored and later used to drive turbines when demand is high or renewable generation is low. This provides a clean, scalable method for grid-scale energy storage.
Advanced Desalination Technologies
Desalination, the process of removing salt from seawater, is energy-intensive. Many existing thermal desalination methods, such as multi-stage flash distillation, rely heavily on boiling and condensing water. Future innovations will focus on highly efficient heat recovery and integration with renewable energy sources. Technologies like membrane distillation, which leverages vapor pressure differences driven by temperature gradients (and thus, latent heat), are being refined to reduce energy consumption and improve cost-effectiveness, offering solutions to global water scarcity.

Smart Systems and IoT for Thermal Management
The integration of smart sensors, Artificial Intelligence (AI), and the Internet of Things (IoT) is revolutionizing thermal management. These technologies can monitor temperatures, pressures, and flow rates in real-time, allowing for predictive maintenance, dynamic optimization of cooling/heating cycles, and proactive fault detection. For instance, in a data center, AI-driven algorithms can adjust cooling tower fan speeds or chiller setpoints based on real-time heat loads and external weather conditions, maximizing the utilization of water’s heat of vaporization for optimal efficiency and minimizing energy waste. Such intelligent systems represent the future of harnessing water’s thermophysical properties for precise and adaptive technological applications.
In conclusion, the heat of vaporization of water, a seemingly simple scientific concept, is a powerful determinant in countless technological processes and systems. Its unique ability to absorb and release significant energy during phase change makes water an unparalleled medium for heat transfer, energy storage, and power generation. As technology continues to evolve, understanding and ingeniously applying this fundamental property will remain paramount for engineers and innovators striving for greater efficiency, sustainability, and groundbreaking solutions to the world’s most pressing challenges.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.