White vinegar, a ubiquitous presence in kitchens worldwide, is more than just a culinary ingredient or a cleaning powerhouse. Its efficacy in both realms stems directly from a fundamental chemical property: its acidity. Understanding the pH of white vinegar is key to unlocking its full potential, whether you’re perfecting a vinaigrette, tackling a stubborn stain, or even exploring its surprising applications in the digital age.
While the title “What is the pH of White Vinegar?” might initially suggest a purely scientific inquiry, its relevance extends far beyond the laboratory. In the context of our website’s core pillars – Tech, Brand, and Money – the seemingly simple acidity of vinegar finds unexpected connections. We’ll explore how this common household item’s characteristics can inform our understanding of digital processes, marketing strategies, and even personal financial management.
The Chemistry of Acidity: Understanding pH and White Vinegar
At its heart, white vinegar is a diluted solution of acetic acid in water. Acetic acid, with the chemical formula CH₃COOH, is a weak organic acid responsible for vinegar’s characteristic sour taste and pungent aroma. The “white” in white vinegar typically refers to its clear color, achieved through a distillation process that removes impurities and colorants often found in darker vinegars like apple cider or balsamic.
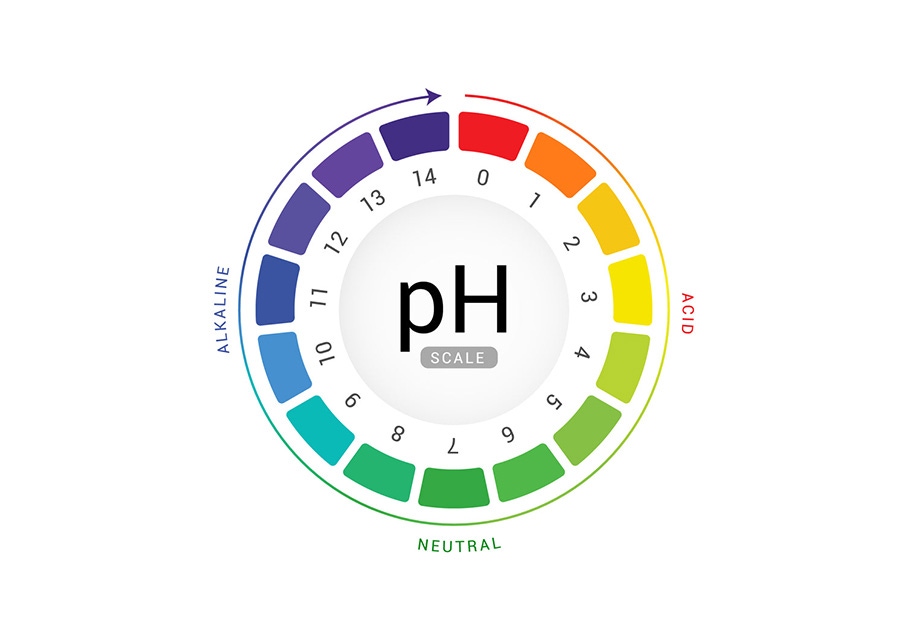
What is pH? A Measure of Acidity and Alkalinity
The pH scale is a logarithmic scale that measures the concentration of hydrogen ions (H⁺) in a solution. It ranges from 0 to 14, with:
- pH 7: Considered neutral. Pure water at room temperature has a pH of 7.
- pH less than 7: Indicates an acidic solution. The lower the pH, the stronger the acid.
- pH greater than 7: Indicates an alkaline or basic solution. The higher the pH, the stronger the base.
Think of it like a Richter scale for acidity and alkalinity. A small change in pH represents a significant change in the concentration of hydrogen ions. For instance, a solution with a pH of 3 is ten times more acidic than a solution with a pH of 4, and one hundred times more acidic than a solution with a pH of 5.
The pH of White Vinegar: A Consistent Acidity
The standard concentration of acetic acid in commercially available white vinegar is typically between 4% and 7%. This concentration directly influences its pH. Most common white vinegars fall within a pH range of 2.0 to 3.5. This makes white vinegar a moderately strong acid, significantly more acidic than, say, milk (pH around 6.5) or even lemon juice (pH around 2.0 to 2.4).
This consistent acidity is precisely why white vinegar is so effective in its diverse applications:
- Culinary: It contributes to the tangy flavor of dressings and marinades, acts as a tenderizer for meats, and can be used in pickling to inhibit bacterial growth.
- Cleaning: Its acidity breaks down grease, grime, mineral deposits (like limescale), and neutralizes odors.
- Gardening: It can be used to lower the pH of soil for acid-loving plants.
The predictable and measurable acidity of white vinegar, quantified by its pH, makes it a reliable tool for a multitude of tasks. This reliability, as we’ll see, has parallels in the digital and branding worlds.
Beyond the Kitchen: Vinegar’s pH and Its Relevance to Tech, Brand, and Money

While the chemical makeup of white vinegar might seem far removed from the fast-paced world of technology, the principles governing its behavior offer insightful analogies. Understanding the consistent and measurable properties of a fundamental element like vinegar can inform how we approach more complex systems.
Tech: The Predictability of Algorithms and Data Sanitization
In the realm of technology, the concept of a predictable and measurable input leading to a consistent output is paramount. This is the bedrock of algorithms and software development. Just as the pH of white vinegar is a reliable indicator of its acidic strength, the output of a well-defined algorithm is predictable given a specific input.
- Data Sanitization and Validation: In digital security and data management, we often need to “sanitize” or validate data. This involves ensuring that data conforms to specific standards and removing any potentially harmful or malformed elements. Think of how vinegar’s acidity can “clean” surfaces by breaking down undesirable substances. Similarly, in programming, specific functions are designed to clean and validate user inputs to prevent errors or security vulnerabilities. For example, a web application might check if a user-provided email address follows a standard format, much like we expect vinegar to consistently have a pH between 2.0 and 3.5.
- Software Functionality: The predictable chemical reaction of acetic acid in vinegar is analogous to the predictable execution of code. Developers rely on consistent behavior from programming languages and libraries to build functional applications. A bug in code is akin to an unexpected chemical reaction – a deviation from the expected and desired outcome.
- AI and Machine Learning: While AI often deals with complex probabilistic models, the underlying datasets often need to be clean and structured. The process of preparing data for machine learning can involve “sanitizing” it, removing outliers or inconsistencies. This mirrors the cleaning and purifying actions of vinegar. Understanding the “acidity” (or specific characteristics) of data is crucial for training effective AI models.
The predictability and measurable nature of white vinegar’s pH provide a simple yet powerful illustration of the importance of well-defined properties and processes in technology.
Brand: Consistency, Reputation, and the “Acid Test”
A brand’s identity and reputation are built on consistency and reliability. Just as consumers expect white vinegar to be consistently acidic, customers expect a brand to deliver on its promises and maintain a recognizable identity.
- Brand Identity and Positioning: A brand’s “pH” can be thought of as its core essence – its values, its mission, and its unique selling proposition. Just as vinegar’s acidity is its defining characteristic, a brand’s defining attributes are what set it apart. A strong brand has a clear and consistent “pH” that resonates with its target audience. Inconsistent messaging or a shifting brand identity can confuse consumers and erode trust.
- The “Acid Test” of Reputation: In marketing and public relations, we often refer to the “acid test” of a brand’s reputation. This refers to critical moments or challenges that reveal the true strength and resilience of a brand. A brand that has consistently upheld its values and delivered on its promises will likely weather these storms more effectively, much like a stable chemical compound resists degradation. The acidity of vinegar, while corrosive to some substances, is predictable and manageable; a damaged brand reputation can be far more difficult to repair.
- Product Development and Quality Control: Just as manufacturers ensure the pH of white vinegar falls within a specific range for optimal performance, brands must maintain rigorous quality control over their products and services. Inconsistent quality can quickly damage a brand’s perception, leading to negative reviews and a decline in customer loyalty. The measurable acidity of vinegar serves as a benchmark for quality; similarly, brands establish benchmarks for their offerings.
The reliable and predictable nature of vinegar’s pH highlights the critical importance of consistency and integrity in building a strong and enduring brand.
Money: Financial Acidity and Investment Risk
The financial world, much like chemistry, involves understanding risks, returns, and the underlying properties of assets. While not a direct chemical comparison, the concept of “acidity” can be metaphorically applied to financial concepts.
- Risk Assessment and Volatility: In finance, “volatility” is a measure of the fluctuation in the price of an asset. Highly volatile assets can be seen as having a certain “acidity” – they are more prone to rapid and significant swings, making them potentially riskier. Just as the pH scale quantifies acidity, financial metrics like standard deviation quantify volatility. Investors need to understand the “acidity” of their investments to make informed decisions.
- Diversification and Balance: In investing, diversification is key to mitigating risk. Spreading investments across different asset classes is like creating a balanced solution. Too much of one “acidic” (volatile) asset can make a portfolio unstable. Similarly, in personal finance, managing cash flow and debt is crucial. Excessive debt can be metaphorically seen as a “financial acidity” that can erode financial health.
- Financial Planning and Budgeting: A solid financial plan is built on understanding your current financial “pH” – your income, expenses, assets, and liabilities. Budgeting helps maintain financial “neutrality” or a healthy surplus, preventing the “acidity” of debt from taking over. Just as you wouldn’t use a highly acidic substance without understanding its properties, you shouldn’t manage your finances without a clear understanding of your financial situation.
The predictable nature of chemical reactions, exemplified by vinegar’s pH, underscores the value of understanding and managing the inherent “properties” and “risks” within financial systems to achieve stability and growth.

Conclusion: The Enduring Relevance of Simple Chemistry
The humble bottle of white vinegar, with its measurable and predictable pH, offers a surprisingly rich tapestry of lessons that extend far beyond the kitchen. From the intricate workings of technology and the strategic construction of brands to the complex landscape of personal finance, the principles of consistency, predictability, and measurable properties remain fundamental.
By understanding that white vinegar typically has a pH between 2.0 and 3.5, we gain not just scientific knowledge, but also a deeper appreciation for how fundamental characteristics drive function and influence outcomes across diverse domains. Whether you’re debugging code, crafting a compelling marketing campaign, or meticulously planning your financial future, the science behind everyday objects like white vinegar can serve as a valuable, albeit metaphorical, guide. The next time you reach for that ubiquitous bottle, remember the profound lessons it holds about the power of understanding underlying principles.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.