In an era defined by rapid technological advancement, understanding fundamental scientific principles often illuminates the hidden mechanisms behind our most sophisticated innovations. Among these, the concept of a “half-life” for a radioactive isotope stands out as a cornerstone, particularly within the realm of technology. Far from being an abstract scientific curiosity, half-life is a critical parameter that dictates everything from the lifespan of medical diagnostics and the efficiency of nuclear power plants to the safe disposal of atomic waste and the design of deep-space probes. For tech professionals, engineers, and innovators, grasping the nuances of radioactive decay and its half-life is not merely academic; it is essential for responsible design, effective implementation, and strategic foresight in an increasingly nuclear-influenced world.
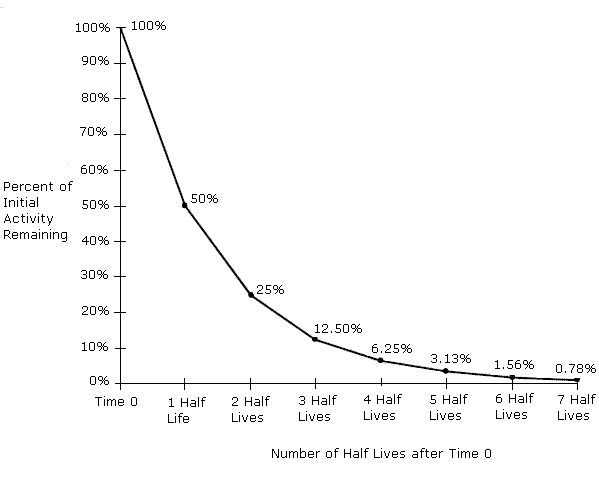
This article delves into the core definition of half-life, then swiftly transitions to explore its profound and multifaceted implications across various technological domains. We will uncover how this intrinsic property of unstable atoms underpins some of humanity’s most transformative technologies, while also presenting significant challenges that demand innovative tech solutions.
The Fundamental Pulse of Decay: Deconstructing Half-Life for Tech Professionals
At its heart, a radioactive isotope is an unstable atomic nucleus that spontaneously transforms into a more stable form, emitting radiation in the process. This phenomenon is known as radioactive decay. The “half-life” is a measure of the time it takes for half of the radioactive atoms in a sample to undergo this decay. It’s a probabilistic process; you can’t predict when a single atom will decay, but for a large collection of identical atoms, the time for half of them to decay is constant and characteristic of that specific isotope.
Decoding Radioactive Decay: The Scientific Backbone
To fully appreciate half-life, we must first briefly touch upon the types of decay that lead to it. Unstable nuclei achieve stability by emitting particles and/or energy. The most common forms of decay include:
- Alpha Decay: The emission of an alpha particle (two protons and two neutrons, identical to a helium nucleus). This significantly reduces the atomic mass and number.
- Beta Decay: The emission of a beta particle (an electron or positron) from the nucleus. This changes a neutron into a proton (or vice versa), altering the atomic number but not the mass significantly.
- Gamma Decay: Often accompanies alpha or beta decay. It involves the emission of high-energy electromagnetic radiation (gamma rays) as the nucleus sheds excess energy, moving from an excited state to a more stable one.
Each radioactive isotope has a unique combination of decay modes and a specific half-life, ranging from fractions of a second (e.g., Polonium-213, with a half-life of 3.7 microseconds) to billions of years (e.g., Uranium-238, with a half-life of 4.46 billion years). This inherent characteristic is not affected by external factors like temperature, pressure, or chemical bonding—a crucial point for its reliability in technological applications.
Mathematical Models and Predictive Power in Tech
While random at the individual atomic level, radioactive decay is predictable for macroscopic samples. This predictability is captured by exponential decay equations. For tech professionals, this translates into powerful predictive capabilities. Engineers can calculate how much radioactive material will remain after a certain period, or how long it will take for a sample to decay to a safe level. This mathematical modeling is vital for:
- Dosimetry in Medicine: Precisely calculating the decaying radioactivity in a patient over time after receiving a radioisotope for diagnostics or therapy.
- Nuclear Reactor Fuel Management: Predicting the depletion of fissile material and the buildup of decay products.
- Waste Management: Estimating the long-term hazard potential of nuclear waste, critical for designing storage solutions.
- Radiometric Dating: Though more of a scientific application, the principles are used in geology and archaeology, sometimes relying on advanced computational tools.
The accuracy of these models is paramount for safety, efficiency, and regulatory compliance across diverse technological sectors.
Distinguishing Between Isotope Lifespans: Short vs. Long Half-Lives
The vast range of half-lives has direct implications for their suitability in different technological applications:
- Short Half-Lives (minutes to days): Ideal for medical diagnostics (e.g., Technetium-99m, 6-hour half-life for imaging; Fluorine-18, 110-minute half-life for PET scans). They provide sufficient radiation for imaging or therapy but decay quickly, minimizing long-term patient exposure and simplifying waste disposal.
- Intermediate Half-Lives (years to decades): Used in industrial gauges, smoke detectors (Americium-241, 432 years), and sometimes in specialized power sources for remote applications. Managing their waste requires careful planning over human timescales.
- Long Half-Lives (thousands to billions of years): Found in nuclear fuels (Uranium-235, Plutonium-239) and long-lived nuclear waste products. These demand extremely robust and long-term solutions for storage and disposal, often spanning geological timescales.
Understanding this distinction is fundamental for selecting the appropriate isotope for a given technological purpose, balancing functionality with safety and environmental impact.
Engineering with Unseen Forces: Half-Life’s Critical Role in Modern Technology
The properties of half-life are not just theoretical concepts; they are actively harnessed and managed across a spectrum of critical technologies that shape our modern world.
Powering the Future: Nuclear Energy and Waste Management Technologies
Nuclear power generation relies directly on the controlled fission of radioactive isotopes, primarily Uranium-235 and Plutonium-239, both with very long half-lives. Understanding their decay rates is crucial for:
- Reactor Design and Fuel Cycles: Engineers calculate fuel enrichment levels, predict the duration of fuel rods, and optimize reactor performance based on the specific half-lives and decay chains of the isotopes involved. This ensures efficient energy production and safe operation.
- Spent Fuel and Waste Management: This is perhaps where half-life poses the greatest technological challenge. The fission products and transuranic elements generated in reactors often have incredibly long half-lives (e.g., Plutonium-239: 24,100 years; Iodine-129: 15.7 million years). Developing technologies for their safe, long-term containment—such as vitrification (incorporating waste into glass matrix), deep geological repositories, and advanced reprocessing techniques to reduce the volume and toxicity of waste—is a monumental undertaking, directly driven by the need to manage materials that will remain hazardous for millennia. Digital modeling and simulation tools are indispensable here for predicting long-term behavior and integrity of storage solutions.
Precision in Medicine: Diagnostic Imaging and Therapeutic Applications
Medical technology has been profoundly revolutionized by the strategic use of radioactive isotopes with carefully selected half-lives.
- Diagnostic Imaging (e.g., PET Scans, SPECT Scans): Positron Emission Tomography (PET) scanners use isotopes like Fluorine-18, with a 110-minute half-life, which is integrated into biologically active molecules (like FDG, a glucose analog). Its short half-life ensures that patients receive a diagnostic dose that decays quickly, minimizing exposure. The technology relies on detecting the gamma rays emitted from positron annihilation, which advanced detectors and software convert into detailed 3D images of metabolic activity in the body.
- Radiation Therapy: For cancer treatment, isotopes like Cobalt-60 (5.27-year half-life) or Iodine-131 (8-day half-life) are used. The specific half-life influences the dosage regimen, treatment duration, and the longevity of the therapeutic source. Precise control over the radiation dose, often guided by real-time imaging and sophisticated treatment planning software, is essential to target cancerous cells while sparing healthy tissue.
- Sterilization of Medical Equipment: Gamma radiation from Cobalt-60 is used to sterilize heat-sensitive medical devices, ensuring product safety without compromising material integrity. The relatively long half-life allows for sustained use of the source.
Industrial Innovation and Beyond: Sensing, Tracing, and Space Exploration
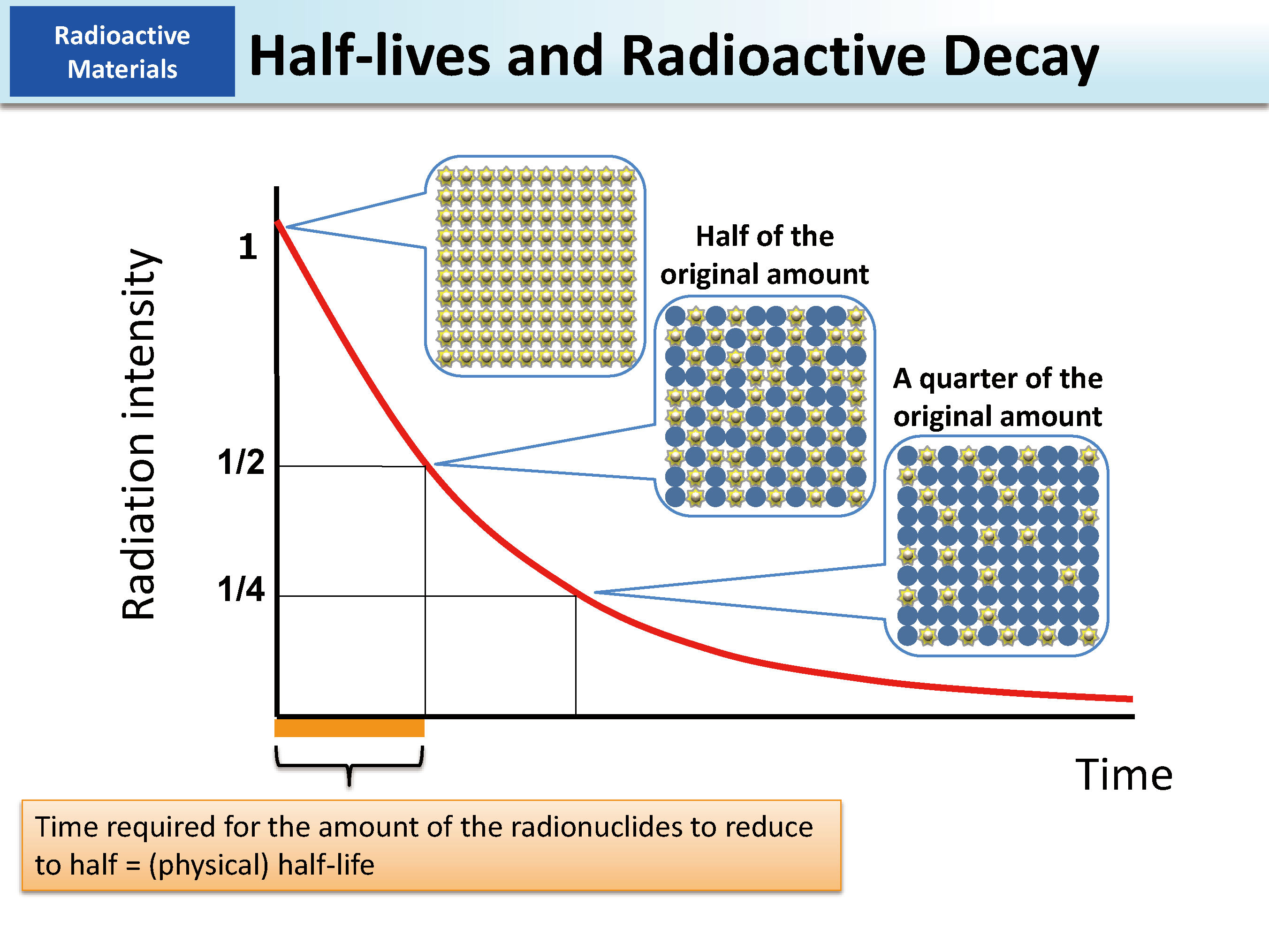
The utility of half-life extends far beyond energy and medicine, permeating various industrial and exploratory technologies.
- Industrial Gauging: Isotopes like Americium-241 are used in industrial gauges to measure the thickness of materials (e.g., paper, plastic films) or the level of liquids in containers. The constant, predictable decay provides a stable radiation source for accurate measurements.
- Smoke Detectors: Many household smoke detectors contain a tiny amount of Americium-241. Its alpha particles ionize the air in a chamber, allowing a small current to flow. Smoke disrupts this current, triggering the alarm. The isotope’s long half-life ensures the device remains operational for many years.
- Radioisotope Thermoelectric Generators (RTGs): For missions to the outer solar system or remote terrestrial applications where solar power is insufficient, RTGs provide reliable electrical power. They use the heat generated by the natural decay of a radioactive isotope (e.g., Plutonium-238, with an 87.7-year half-life) to produce electricity. The half-life is critical for determining the lifespan of the power source and the energy output over the mission’s duration.
Managing the Invisible: Digital Security, Safety, and Ethical Tech Considerations
The power and ubiquity of radioactive isotopes necessitate robust management strategies, where technology plays a pivotal role in ensuring security, safety, and ethical governance.
Securing Nuclear Infrastructure: Cybersecurity Challenges
Facilities handling radioactive materials, especially nuclear power plants, are prime targets for cyberattacks. A thorough understanding of half-life informs:
- Control System Design: Cybersecurity measures must protect the digital control systems that manage reactors, waste storage, and material handling. This includes intrusion detection, robust firewalls, and air-gapped networks.
- Data Integrity: Ensuring the integrity of data related to isotope inventories, decay rates, and safety parameters is critical to prevent malicious manipulation or accidental errors that could have catastrophic consequences.
- Physical-Digital Integration: The convergence of physical security (e.g., radiation monitors, access controls) with digital security systems creates complex challenges requiring integrated technological solutions.
Regulatory Frameworks and Advanced Monitoring Technologies
International and national regulatory bodies (like the IAEA) set stringent standards for the handling, transport, and disposal of radioactive materials, all underpinned by the concept of half-life.
- Monitoring Technologies: Advanced sensors, drone-based radiation mapping, and remote monitoring systems leverage digital technologies to track radioactive sources, detect leaks, and ensure compliance with safety protocols. These systems often employ AI and machine learning for anomaly detection and predictive maintenance.
- Traceability and Inventory Management: Sophisticated software systems are used to maintain precise inventories of radioactive isotopes, tracking their half-life decay to ensure accountability and prevent illicit trafficking.
The Ethical Compass: Balancing Innovation with Responsibility
The applications of half-life-dependent technologies raise significant ethical questions. Tech professionals are at the forefront of developing solutions that ensure public safety, environmental protection, and non-proliferation.
- Dual-Use Technologies: Many nuclear technologies have both peaceful and military applications. Ethical considerations guide the development of safeguards and monitoring tools to prevent the misuse of isotopes.
- Public Perception and Transparency: Transparent communication and public engagement, often facilitated by digital platforms and data visualization tools, are essential to build trust and address concerns about nuclear technologies.
- Long-Term Environmental Stewardship: The challenge of managing long-lived nuclear waste for geological timescales demands ongoing technological innovation to ensure that future generations are not unduly burdened.
The Next Frontier: Half-Life in Emerging Tech and Research
As technology continues its relentless march forward, the understanding and manipulation of half-life remain a fertile ground for new discoveries and applications.
Advanced Materials Science: Doping and Radiation Hardening
In materials science, knowledge of half-life is critical for:
- Semiconductor Doping: While not directly using radioactive isotopes for doping, understanding decay processes helps in developing stable and effective dopants. More directly, the effects of radiation on materials (radiation hardening) are critical for electronics operating in high-radiation environments (e.g., space, nuclear facilities). New materials are being engineered to resist radiation damage, often informed by how radiation interacts with atomic structures over time.
- Novel Materials for Energy Storage: Researchers are exploring new battery technologies, some of which might involve long-lived isotopes for sustained energy output, requiring careful half-life considerations for safety and device longevity.
AI and Machine Learning in Isotope Management and Decay Prediction
Artificial intelligence and machine learning are increasingly being applied to optimize and manage systems involving radioactive isotopes:
- Predictive Maintenance: AI algorithms can analyze sensor data from nuclear reactors to predict equipment failures, optimize fuel cycles, and enhance safety protocols, all indirectly linked to the ongoing decay processes.
- Medical Isotope Production Optimization: ML can optimize the cyclotron or reactor conditions for producing medical isotopes with specific half-lives, ensuring efficient and timely delivery to hospitals.
- Simulating Long-Term Waste Behavior: AI-powered simulations can model the complex geochemical interactions and decay processes within geological repositories over thousands of years, providing invaluable data for design and risk assessment.
Exploring Exotic Isotopes for Novel Energy Solutions and Quantum Applications
Future research into exotic isotopes and their decay properties could unlock entirely new technological paradigms:
- Next-Generation Nuclear Fission/Fusion: Understanding the half-lives of new, experimentally produced isotopes is crucial for developing advanced nuclear reactor designs, including small modular reactors (SMRs) and fusion reactors, which aim for greater safety and efficiency.
- Quantum Computing: While speculative, the precise control and understanding of atomic nuclei, including their decay characteristics, might find esoteric applications in future quantum technologies or advanced sensing, where fundamental properties of matter are exploited at their most basic level. The quest for ultra-stable quantum bits could intersect with the stability and decay properties of specific isotopes.

Conclusion: The Enduring Relevance of Half-Life in a Technologically Driven World
The concept of a half-life, the immutable clock governing radioactive decay, is far more than a scientific footnote; it is a fundamental principle woven into the fabric of modern technology. From powering our cities and healing our bodies to exploring the cosmos and securing our infrastructure, a deep understanding of half-life enables engineers, scientists, and tech innovators to harness atomic forces responsibly and effectively. As we push the boundaries of what’s possible, the challenges and opportunities presented by radioactive isotopes, and the precise timing of their decay, will continue to drive technological progress, demanding ever more sophisticated solutions in safety, security, and sustainability. For anyone operating at the cutting edge of tech, the half-life of a radioactive isotope remains a concept of enduring and critical relevance.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.